HKSTP Incubatee ImmunoDiagnostics Receives European CE Mark Approval for Its High Throughput COVID-19 Diagnostic Products
ImmunoDiagnostics’ sophisticated diagnostics R&D a testament to Hong Kong’s rise as a global biomedical technology hub


Dr. Kelsey Zhong, Deputy Executive Director, ImmunoDiagnostics Ltd. (3rd from right) and the research team present three diagnostics products for the COVID-19 virus.
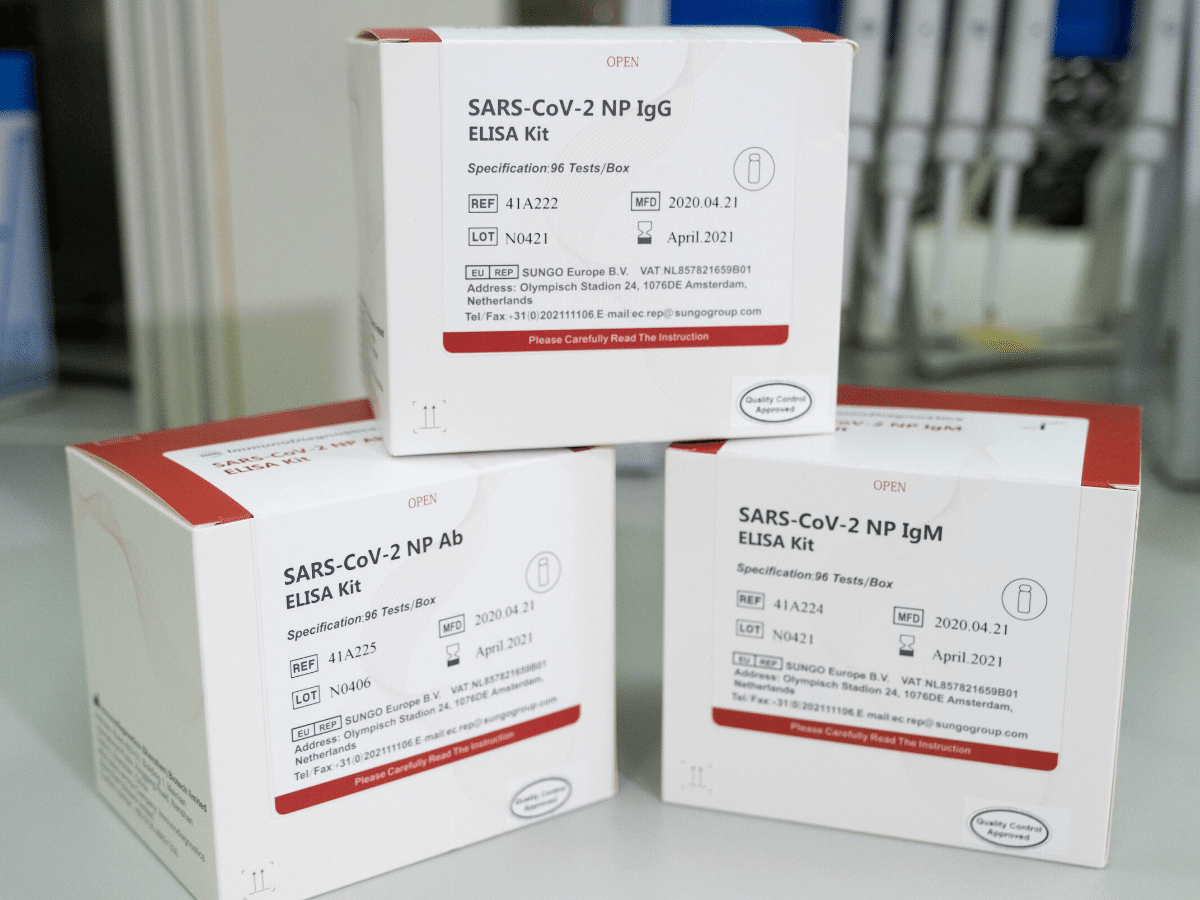
The diagnostics products for the COVID-19 virus developed by ImmunoDiagnostics have received the European Union’s CE certification, and have been delivered to several European countries for clinical use.
Hong Kong, 18 May, 2020 – Hong Kong biotech start-up ImmunoDiagnostics Ltd. announced it has received the European Union’s CE certification for three of its COVID-19 rapid testing diagnostics products. The certification enables the three CE-marked antibody detection kits to be used for diagnostic applications across all EU countries, as well as in other markets including Malaysia, South Korea, Thailand, Iran, and Turkey.
ImmunoDiagnostics is a spin-off from the University of Hong Kong and is a current incubatee under Hong Kong Science and Technology Parks Corporation’s (HKSTP) Incu-Bio Programme. The company is dedicated to biomarker discovery, assay development and In-Vitro Diagnostics (IVD) of major chronic diseases and infectious diseases. In particular, the company specialises in cardio-metabolic diseases, autoimmune diseases, and emergent pathogens such as COVID-19.
ImmunoDiagnostics’ COVID-19 test kit series is designed to boost the critical test capacity for health systems around the world in combating the novel coronavirus by determining the circulating antibodies against the virus in human blood, with a clinically validated high specificity (>90%) and sensitivity (>92%). It offers a high-throughput, cost-effective approach in delivering 90 tests per assay kit with results available within 2.5 hours.
It also provides a high degree of compatibility with many current testing systems, as its absorbance plate reader is commonly used and easily accessible in many diagnostic and analytical labs. The assays utilise the ELISA (enzyme linked immunosorbent assays) platform, which is reliable, stable and one of the most sensitive immunoassay platforms available today. During operation, operators can avoid contact with any highly contagious specimens such as saliva and sputum, instead testing serum or plasma samples, providing an extra level of safety for them.
With the CE certification received, the ImmunoDiagnostics products have entered mass production. Test kits for over 300,000 people have already been delivered to several European countries including Italy, Spain, Czech Republic, Austria and Switzerland. Requests from other markets have been received as well.
“We are proud to see ImmunoDiagnostics’ hard work and research become commercially viable products that can make a difference in the fight against the COVID-19 pandemic, not only in Hong Kong but across the globe,” said Dr. Kelsey Zhong, Deputy Executive Director, ImmunoDiagnostics Ltd. “This marks a major milestone in our journey to become the largest and most reliable supplier of in-vitro diagnostic reagents in Asia Pacific, as well as a global pioneer in the production of research products related to immuno-metabolism. We will continue to dedicate our research efforts on biomarker discover, assay development and in vitro diagnostics of major chronic and infectious diseases for the health of the global community.”
Professor W. John Kao, Head of the Biomedical Technology Cluster, HKSTP, added, “We are delighted to see the growing global impact of Hong Kong’s capability in biomedical technologies, with the EU’s recognition of ImmunoDiagnostics’ technology developed right here at Hong Kong Science Park. It reflects the high international standard of Hong Kong’s biotech R&D and showcases of our contribution towards the battle against the COVID-19 pandemic. Hong Kong is strategically positioned to be a world-class biotech research hub with our solid IP protections, strong pool of research professionals, and globally recognised clinical trial capabilities. HKSTP will continue to provide a comprehensive support to the local biotech community to drive R&D breakthroughs for the greater good.”
In addition to the CE-certified products, ImmunoDiagnostics also developed a comprehensive range of COVID-19 related products for research purposes, that are already being used by many top institutes globally, including National Institutes of Health of United States, University of Washington, University of Miami and John Hopkins University.
###
For media enquiries, please contact pr@hkstp.org. Explore our latest news in the PRESS ROOM.

